
The letter Z is used to represent an atomic number.Ītomic mass cannot be used to determine the element’s kind. It is the total number of protons in the nucleus of an atom. The number of protons in an element’s nucleus is generally represented by its atomic number. 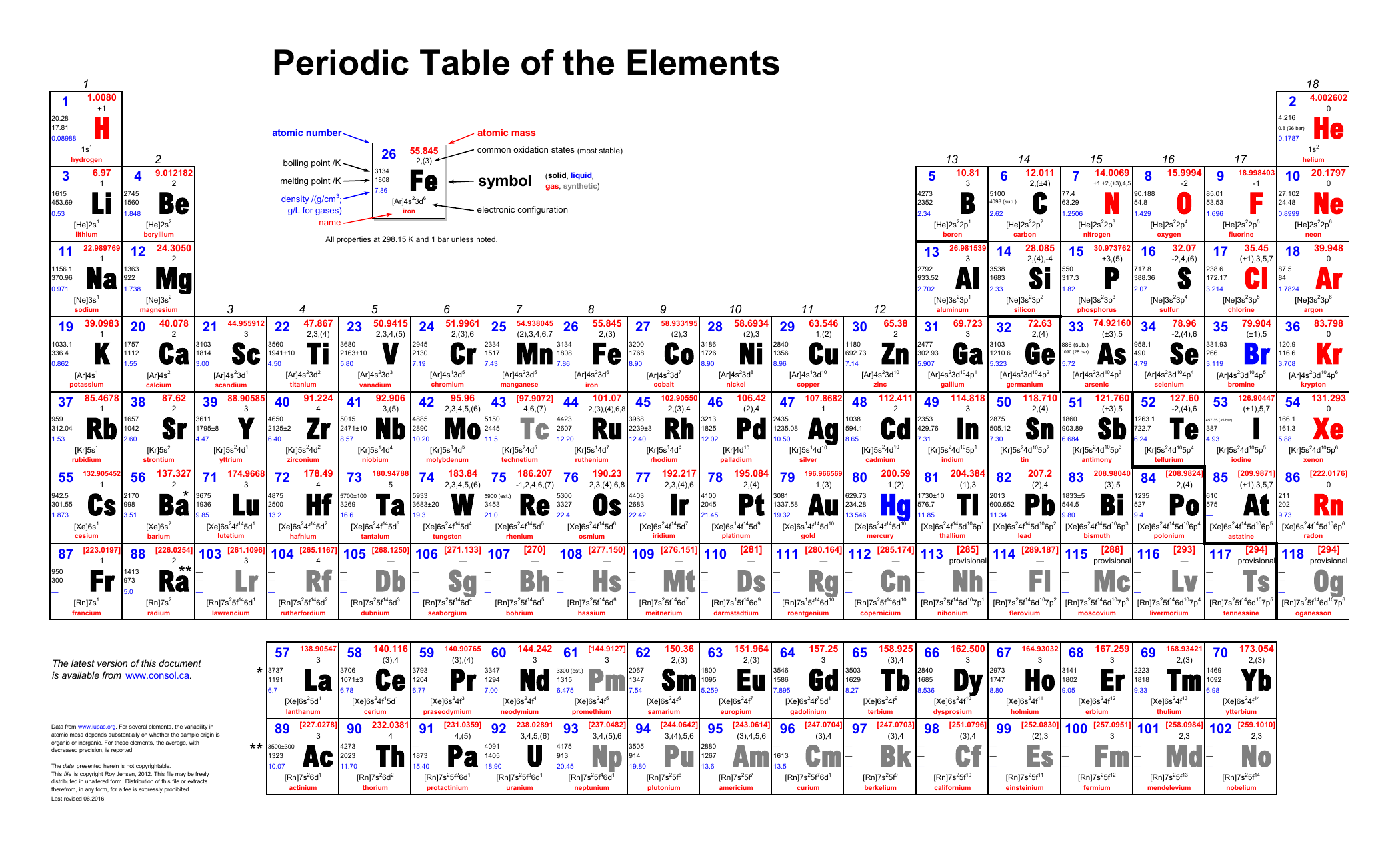
Difference Between Mass Number and Atomic NumberĪtomic mass is proportional to the number of neutrons and protons contained in an element’s nucleus. Where A = Mass number, Z = Atomic number and n= no. The Relationship Between Mass Number and Atomic Number is stated as: Relationship Between Mass Number and Atomic Number If the element is X, then the atomic number Z and mass number A are written in chemical notation as. Notation of the Atomic Number and Mass Number Because the mass of the sodium atom is 23, we may state that A=23 for sodium. The letter A represents an element’s mass number. As a result, the mass number of hydrogen is only one. Similarly, because a carbon atom contains 6 protons and 6 neutrons, its mass number is 6+6=12.Ī hydrogen atom has one proton but no neutrons. One atom of sodium, for example, contains 12 neutrons and 11 protons, therefore the mass number of sodium is 11+12=23. Mass number = Number of protons + Number of neutrons The mass number is equal to the sum of the number of protons and neutrons or, The mass number of an element is the total number of protons and neutrons contained in one atom. Because electrons have a tiny mass in comparison to protons and neutrons, the mass of an atom is determined solely by protons and neutrons. Protons, neutrons, and electrons make up an atom. As a result, the atomic number of an element does not change throughout a chemical reaction and stays constant. The number of electrons can change throughout a chemical reaction, while the amount of protons does not. Chemical processes solely involve the electrons of atoms, not their protons. An ion, on the other hand, is formed by either removing or adding electrons to a conventional atom, and therefore contains either fewer or more electrons than protons. Only a neutral atom has an equal number of protons and electrons an element’s atomic number is equal to the number of electrons in the neutral atom, not the ion. As a result, we may state-Ītomic number of an element = Number of electrons in one neutral atom The number of protons in a typical atom is equal to the number of electrons. Because different elements have distinct atomic numbers, their atoms differ. Thus, the atomic number differentiates one element’s atoms from the atoms of another element. 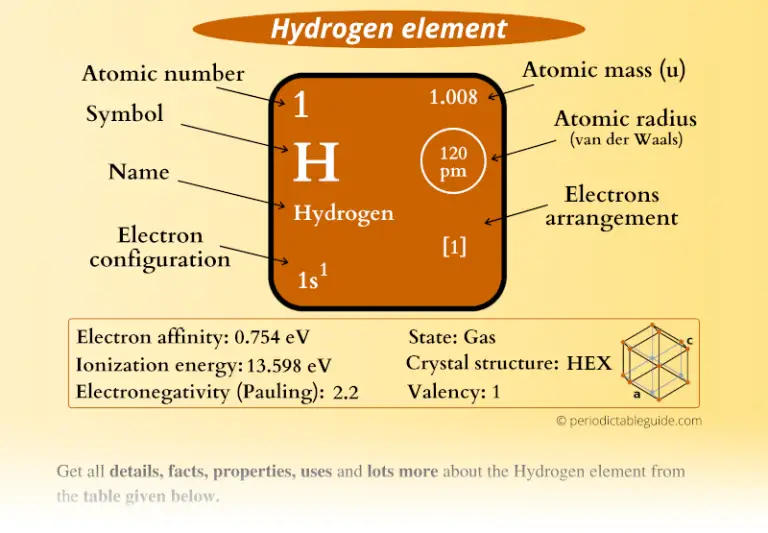
There is no other element with an atomic number of 6.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
